 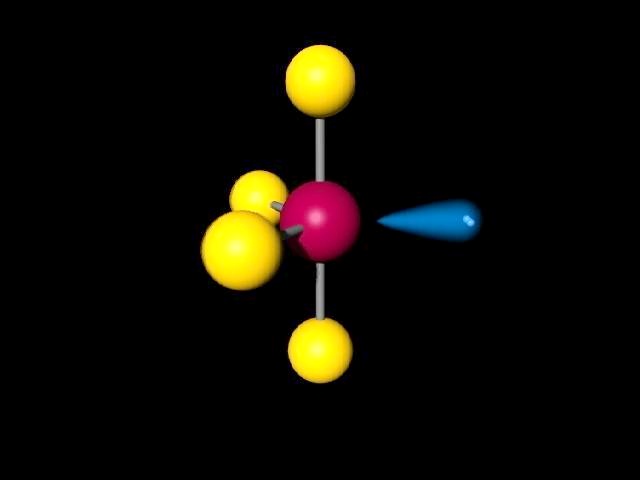
So, that bends the axial fluorines together a bit. The seesaw shape maximizes the bond angles of the single lone pair and the other atoms in the molecule. The bond angles for molecules having a tetrahedral geometry are generally 109.5 degrees, but as there are double bonds, it might be close to this angle but not precisely 109.5 degrees. 
Note though that the structure is distorted a bit due to the repulsive forces of the lone pair of electrons you see (not bonded). With one lone pair of valence electrons, you get a seesaw molecular geometry. The See-Saw shape is basically the same shape as the Trigonal Bipyramidal except one bond is being removed from it. Using this information, we can determine the molecular geometry for. The See-Saw is a molecular shape where there are 4 bonds attached to the central atom along with 1 lone pair. Note through that the structure is distorted a bit due to the repulsive forces. Therefore, you can put 6x4 on each fluorine, 2x4 to account for four single bonds, and 2 for the last 2 valence electrons available.Īs a result, you have 5 electron groups, so the electron geometry would be trigonal bipyramidal. F F 90 F 120 S F FIGURE 8.23 Sulfur tetrafluoride is a molecule with seesaw geometry. With one lone pair of valence electrons, you get a seesaw molecular geometry. A quick explanation of the molecular geometry of SF4 including a description of the SF4 bond angles.It is important to note that you must first draw the corr. 
You can put sulfur in the middle because fluorine tends to make single bonds. An example of a seesaw shaped molecule is. 9.2 (a) 120° (b) If the blue balloon expands, the angle between red and green. However, one of the BAB angles in the seesaw is 120°, so it must be derived from a trigonal bipyramid. Within the context of VSEPR theory, you can count electrons to determine the electron geometry ("parent" geometry). SF4 Molecular Geometry And Bond Angles SF4 molecular geometry is see-saw with one pair of valence electrons. 9 Molecular Geometry and Bonding Theories Visualizing Concepts 9.1 Removing an atom from the equatorial plane of trigonal bipyramid in Figure 9.3. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
